Corporate governance
Corporate Governance continues to be an important area of focus for the Board. The Board believes that good Corporate Governance is essential for the long-term success of the business and this is ultimately the responsibility of the Board and its Committees.
Further information regarding governance at OXB can be found in our 2025 Annual Report.
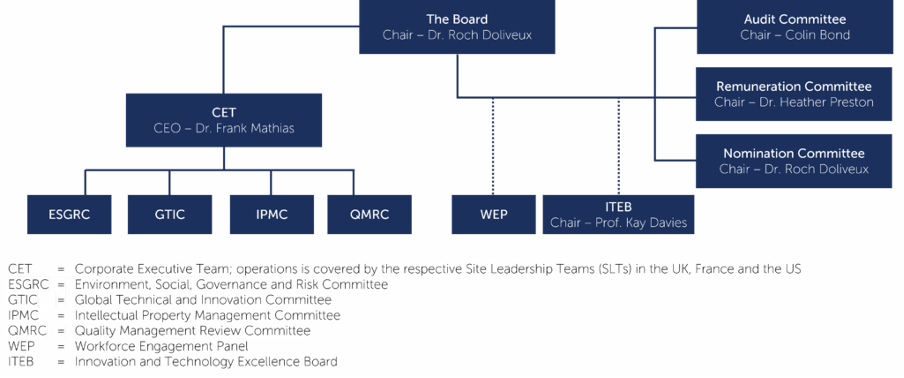
The Board
The Board is collectively responsible for promoting the success of the Group by directing and supervising the Group’s activities to create shareholder value. In doing so, it ensures that there are robust corporate governance and risk management processes in place.
The Board also maintains oversight of Innovation, Quality, Health and Safety, ESG and Risk Management. The respective management leads of these areas prepare reports for every Board meeting.
Details of the matters reserved to the Board, Role of the Chair, the Vice Chair, the Chief Executive Officer and the Senior Independent Director can be accessed via the following links:
Board Committees
Certain responsibilities are delegated to three Board Committees – the Audit, Nomination and Remuneration Committees. In addition, the Company also has an advisory committee, the Innovation and Technology Excellence Board. These Committees operate under clearly defined terms of reference.
The Group also has an established WEP comprising employees from all levels and functions across the Group.
Remuneration Committee
The Remuneration Committee comprises of four independent Non-Executive Directors: Dr. Heather Preston (Chair), Professor Dame Kay Davies, Mr. Peter Soelkner and the Company Chair, Dr. Roch Doliveux.
The Chair of the Remuneration Committee is available to discuss matters with shareholders throughout the year.
The Company’s policy on remuneration is to attract, retain and incentivise the best staff in a manner consistent with the goals of corporate governance. In setting the Company’s remuneration policy, the Remuneration Committee considers a number of factors, including the basic salaries and benefits available to Executive Directors of comparable companies.
Audit Committee
The Audit Committee comprises of three independent Non-Executive Directors: Mr. Colin Bond (Chair), Dr. Heather Preston, and Peter Soelkner.
On behalf of the Board, the Audit Committee oversees the risk management strategy and appetite, the appropriateness and effectiveness of internal control processes, and Corporate Governance Code compliance.
Nomination Committee
The Nomination Committee comprises of four independent Non-Executive Directors: Dr. Heather Preston, Professor Dame Kay Davies, Mr. Peter Soelkner and the Company Chair Dr. Roch Doliveux, who is also Chair of the Nomination Committee. The Nomination Committee evaluates the balance of skills, knowledge and experience on the Board and, in the light of this evaluation, determines the role and capabilities required for particular appointments.
Innovation and Technology Excellence Board
The Innovation and Technology Excellence Board (ITEB) is a committee made up of external advisers and CET members. The Committee is chaired by the Non-Executive Director Professor Dame Kay Davies. Whilst not a formal Board Committee, the committee reviews and assesses technical and process developments in the field of cell and gene therapy and regularly reports to the Board.
Diversity and inclusion
Corporate executive team
The CET is responsible for the global management of the Group. The CET meets on a bi-weekly basis, with the Site Heads joining every other CET meeting. Operations are covered by the respective Site Leadership Teams (SLTs) in Bedford, MA, US, Lyon and Strasbourg, France and Oxford, UK.
There are four CET sub-committees covering the major business operational areas. These sub-committees meet on a regular basis and are attended by certain CET members and other relevant senior managers from the business.
The CET sub-committees are:
- Environment, Social, Governance and Risk Committee (ESGRC) – this sub-committee combines ESG and Risk Management Committees comprising senior managers from all parts of the business across all OXB sites.
- Global Technical and Innovation Committee (GTIC) – this sub-committee is authorised by the CET to review all technical and innovation activities associated with the Group’s capabilities, platform technologies and technical innovations across all OXB sites. It is the primary forum for discussing new projects related to the technology / innovation roadmap and making strategic and budgetary decisions on the best uses of OXB resources.
- Intellectual Property Management Committee (IPMC) – this sub-committee comprises senior members of technical and IP teams and is responsible for ensuring the protection of Intellectual Property across all OXB sites.
- Quality Management Review Committee (QMRC) – this sub-committee provides global oversight in relation to quality and compliance across all OXB sites and is supported by more frequent location/site-specific quality forums where each of the sites review quality related KPIs, compliance, etc. to evaluate the overall health of the Quality Management System at the site-level. Within their area of responsibility these sub-committees set objectives and targets, monitor performance against KPI’s, ensure compliance with GxP and other relevant requirements and monitor expenditure against budget and risk management. Important matters arising from all of these sub-committees are referred to the CET.
Governance and compliance of subsidiaries
Defending ethical values
Our position as a manufacturer of medicines and as a player in the protection and improvement of health, our expertise in the safety of medicines and our international activity impose that we always act in accordance with legal requirements, implementing high standards of integrity and ethics through our Ethics and Compliance Programme, and to behave as a socially responsible company.
Our staff can sometimes find themselves in confusing situations in the course of their work. Our Code of Conduct was developed to set up rules of conduct and integrity which constitute a necessary reference for each of us, to help us make the right decisions, at the right time, in an increasingly complex professional environment.
Code of conduct:
Oxford Biomedica’s solid reputation is based on values passed on from one generation to the next.The purpose of this Code is to establish the principles of compliance and ethics that apply to the company and to individual officers, directors, employees, customers and contractors such as suppliers, service providers.
Sharing your concerns:
We are strongly committed to fighting against all unethical behaviours, corrupt practices, fraud, collusion, and discrimination. Employees having concerns or doubts regarding any behaviour that may violate the code are encouraged and required to address their concerns to their director, manager, Human Resources Department or any member of the Leadership Team.
Protecting personal data and ensuring privacy
OXB France implements a strict confidentiality and security policy aimed at protecting the personal data it processes, and deploys an appropriate training programme to raise awareness of the staff on these regulations.
Protection of personal data and commitments to confidentiality and security:
This personal data protection charter (the “Charter”) applies to Oxford Biomedica France, whose registered office is located in Illkirch-Graffenstaden (67400), France, as well as Lyon (69002).
In the context of this Charter, your personal information is referred to as personal data (“Personal Data”). We may also sometimes refer collectively to the management, collection, protection and storage (“the Processing”) of your Personal Data.
This Policy also includes information regarding sharing your Personal Data.
We have set up this Charter so that you can communicate your Personal Data in compliance with a strict confidentiality and security policy by which we undertake to protect your private life and to manage your Personal Data in a fair and transparent manner.
Categories of personal data collected:
We may collect or obtain such Personal Data because you have provided it to us (for example, through a contact form on our website), or through communication by third parties (for example, your employer or third-party service providers that we use for our business purposes) or because such Personal Data is publicly available.
We may also collect or obtain Personal Data about you by noting or deriving such data from the way you interact with us directly or indirectly through other persons (for example, partners, co-controllers or subcontractors, who may be involved in data collection operations on behalf of Oxford Biomedica France).
The Personal Data we collect or obtain may include: first name, last name, email address. In some cases we may also collect other specific categories of Personal Data about you either with your prior explicit consent, or under contract, or if we are required to do so by law.
Nature and purpose of the processing:
We will use your Personal Data to inform you about the services we offer and the management of activities.
If you no longer wish to receive commercial solicitations from us, you may say so by sending a request on the alert platform using the following address: OxfordBiomedicaFR-compliance@oxb.com
Recipients of the personal data:
In the context of one or more of the above-mentioned Processing purposes, we may communicate certain information to Oxford Biomedica France.
We may also be required to disclose your Personal Data in the event that such disclosure is required by law, an administrative authority or for the purposes of legal proceedings.
Protection of personal data:
Oxford Biomedica France takes all necessary precautions, including administrative, technical, organisational, and physical measures, to protect the Personal Data of employees, customers and partners against loss, theft and fraudulent removal, as well as against unauthorised access, disclosure, alteration or destruction of Personal Data.
Although we apply all security measures deemed reasonably necessary once your Personal Information has been collected, the transmission of data over the Internet (including by e-mail) is never completely secure. We strive to protect the Personal Information in our possession, but we cannot guarantee the security of the communication of data transmitted by us or to us.
Principles applicable to the processing of personal data:
Oxford Biomedica France endeavours to ensure that the Personal Data in its possession is:
-
Collected for specified, explicit and legitimate purposes and not further processed in ways incompatible with those purposes;
-
Processed fairly and in accordance with the law, in full transparency to the data subject (individuals);
-
Adequate, relevant and limited to what is necessary for the purposes for which they are processed (data minimisation);
-
Accurate and, where necessary, kept up to date;
-
Kept in a form which permits identification of data subjects for no longer than is necessary for the purposes for which they are processed;
-
Processed in such a way as to ensure appropriate security;
-
Transferred from one country to another only subject to (i) an adequacy finding by the European Commission or (ii) appropriate safeguard measures.
Protection of children’s privacy:
Oxford Biomedica France understands the importance of protecting the privacy of children, especially in a digital environment. As such, our site(s) is (are) not intentionally designed for or directed at children under the age of 16. It is our policy never to knowingly collect or maintain information about anyone under the age of 16.
Retention period:
We will retain your Personal Data in our systems for the longest of the following: (i) the period necessary for the activity or operations; (ii) any retention period imposed by law; (iii) the end of the applicable statute of limitations period following any litigation or investigation that may arise in connection with the activity or services concerned, unless a prior request for deletion is sent to the ethical platform. At the end of this period, your Personal Data will be destroyed, in accordance with the applicable regulations.
Rights of individuals:
In accordance with the laws and regulations relating to the protection of personal data, any individual is entitled to exercise the following rights with Oxford Biomedica France, who carries out processing of their Personal Data:
-
To be informed of the existence and purposes of any processing of his/her Personal Data;
-
To access his/her Personal Data and to request the rectification or deletion of his/her Personal Data or a limitation of the Processing;
-
To object to the Processing and to request the portability of his/her Personal Data;
-
To withdraw, at any time, his/her consent given to Oxford Biomedica France;
-
To promptly inform the data protection officer of any loss or fraudulent removal (unlawful processing) of his/her Personal Data.
In order to exercise any of your rights, you may log on to the platform at the following email address: OxfordBiomedicaFR-compliance@oxb.com
Right to complain:
If you are dissatisfied with the way in which we have handled your Personal Data or if there are any questions or requests unanswered by our services, you have the right to file a complaint by the relevant Administrative Authority for Personal Data Protection.
Enhancing the relationship with our third parties
Prevention of Corruption:
Oxford Biomedica France is committed to fight against corruption on a daily basis, both internally and in relation with third parties.
We are committed to establish long-term relationships with its suppliers and service providers, based on a responsible purchasing policy, to ensure mutual success.
In addition to the Code of Conduct, we establish high standards of ethics and integrity for our business partners and suppliers through:
-
a business integrity guide “Ethical Business Practices”,
-
a process for selecting and monitoring third party intermediaries, including a questionnaire, the Third Party Approval Form.
In addition, training and information is provided to employees working with health authorities, suppliers, providers and public or private customers.
Inappropriate behaviour can be reported online using our alert platform: https://oxbfr.alertcenter-ikarian.eu/entreprises.
Ensuring the Quality of our Services
The performance of our quality management systems, backed by an ambitious Quality Policy, enables us to provide our customers with products that meet the strictest regulatory standards.
This is achieved through compliance with good manufacturing practices, the deployment of a demanding quality manual that lives on through all our employees, and the sharing of a common quality culture across all our sites.


