Home – CDMO Solutions | Process Development
Process development services for cell and gene therapy
OXB delivers reliable GMP manufacturing of viral vectors to support your program from first-in-human trials through global commercial launch. Leveraging state-of-the-art facilities, robust processes, and innovative technologies, we provide scalable, high-yield, and regulatory-ready manufacturing for multiple vector types.
Benefit from our rigorous controls: our quality-focused GMP processes and robust analytics ensure comprehensive, end-to-end CMC support, guiding your viral vector product through every stage of development and into commercialization with confidence.
Why choose OXB for viral vector process development?
Our experience and cutting-edge platforms provide measurable advantages:
Reduced process variability
Consistent, reproducible outcomes.
Regulatory-ready design
Built-in quality and compliance.
Optimized yield
Increased productivity and lower vector cost per dose.
Scalable solutions
Smooth transition from clinical to commercial scale.
Speed to clinic
Streamlined workflows minimize development timelines.
Our proven process development approach
Experienced partner for adenovirus development and manufacturing

Serum-free, chemically defined suspension systems
Eliminate animal components for safer, consistent, and scalable viral vector production.

Parameter optimization for high-yield viral vectors
Fine-tune critical process parameters to maximize yield and ensure reproducibility.

Leverage platform approach to reduce time and cost
Streamlined development through proven platform processes minimizes risk, accelerates timelines, and cuts expenses.

End-to-end support from lab to launch
Integrated solutions from process design to tech transfer for faster development timelines.
Process development platforms for viral vectors
Accelerate your therapy development with a tailored process strategy. Contact OXB today to discuss how our viral vector process development services can meet your program’s needs.
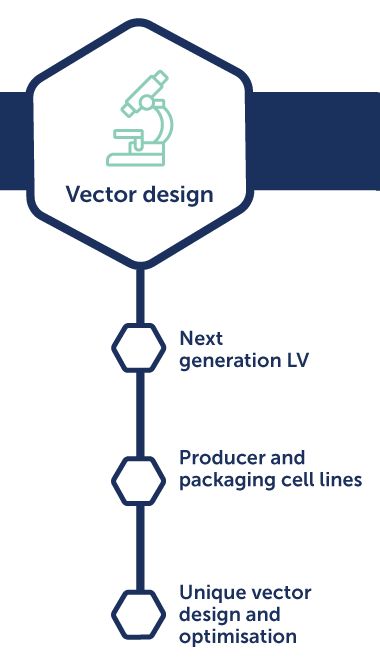
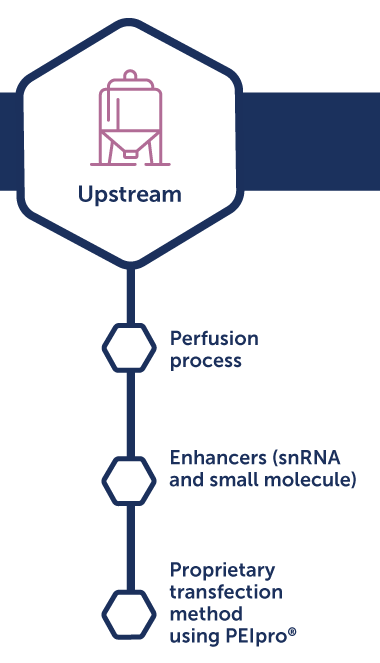
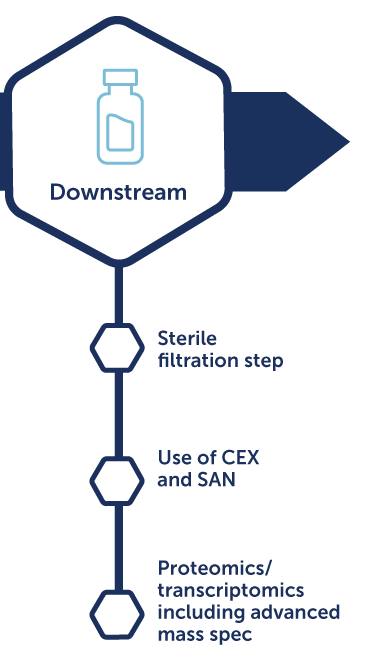
Lentivirus process development
Benefits:
- Increased productivity and quality
- Streamlined process to clinic
- Demonstrated regulatory compliance
- Cost-effective standard solutions
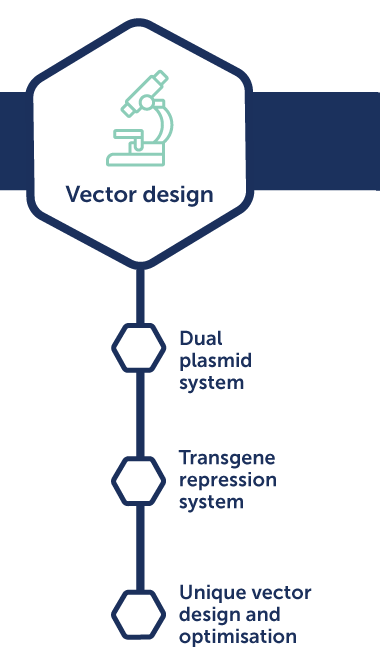
AAV process development
Benefits:
- Maximum productivity and highly potent product
- Faster time to GMP with scalable approach
- Low ratio of empty capsids compared to full capsids
- Improved drug product stability
Partner with OXB for process development excellence
Accelerate your therapy development with a tailored process strategy. Contact OXB today to discuss how our viral vector process development services can meet your program’s needs.

Explore OXB’s other CDMO solutions
Related resources
The TetraVecta™ System: Optimisation & Exemplification of OXB’s Next Generation Lentiviral Vector
View PDF >Enhancing Titres of Therapeutic Viral Vectors using the Transgene Repression in Vector Production TRiP System
This paper describes using a TRiP system to temporarily repress transgene expression during production, boosting viral vector titers for therapeutic use.
View PDF >Frequently asked questions
OXB offers end-to-end viral vector process development services for cell and gene therapy programs, including serum-free suspension process development, parameter optimization, and integrated lab-to-launch support. Our solutions ensure high-yield, reproducible, and scalable processes ready for clinical and commercial applications.
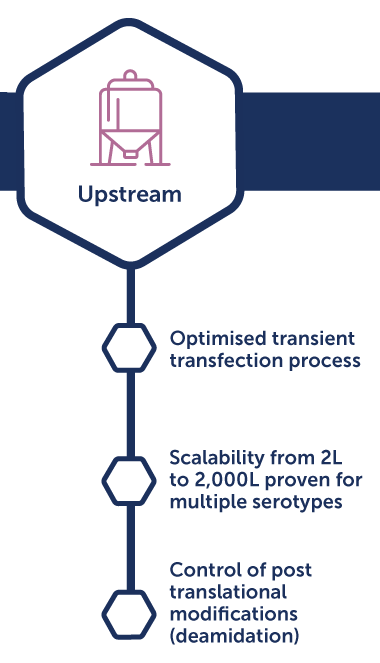
Over the years, OXB has consistently refined its processes to drive higher yields and improved product quality. Our scientists have explored the design space extensively, identifying and optimizing the critical levers that govern productivity and consistency. By focusing on these parameters, OXB delivers viral vector processes that are not only robust and reproducible but also scalable for both clinical and commercial applications.
Yes. Unlike many CDMOs, OXB provides expertise in construct design and optimization to ensure viral vectors are engineered for productivity, potency, and manufacturability. By addressing construct considerations at the earliest stage, we help reduce downstream risks and accelerate successful process development.
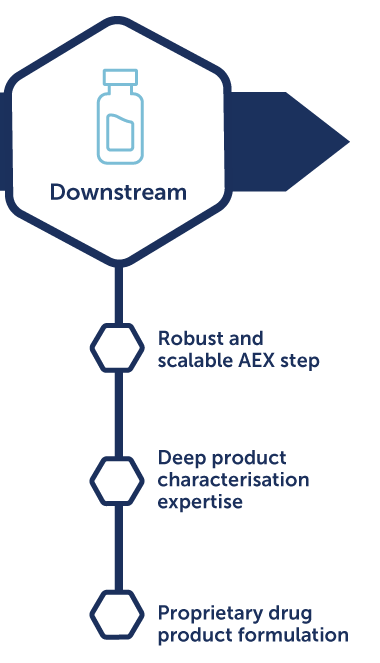
OXB takes an integrated approach to upstream and downstream development. On the upstream side, we optimize cell culture, media, and bioreactor conditions to achieve consistent high yields. Downstream, we innovate in purification and recovery methods to maximize yield and ensure efficiency, scalability, and quality that meets regulatory expectations.
OXB is among the few CDMOs offering dedicated formulation development for viral vectors. Our scientists design and develop formulations for various vectors with considerations to route of delivery that enhance stability, extend shelf life, and preserve potency throughout clinical and commercial supply chains. This capability ensures therapies maintain quality from production to patient administration.
Using our dedicated platforms, LentiVector™ platform for lentivirus and inAAVate™ platform for AAV, OXB delivers tailored process development, streamlined workflows, and regulatory-ready designs. This platform approach accelerates timelines, minimizes risk, and enables scalable, high-quality viral vector production for multiple applications.
OXB’s streamlined process development workflows and platform-based strategies reduce development timelines by enabling rapid optimization, early scalability, and smooth tech transfer to GMP manufacturing. This helps programs move faster from research to clinical readiness with lower risk and cost.
All process development activities are performed with regulatory considerations in mind, including quality-by-design principles, robust analytics, and process documentation. This ensures smooth tech transfer to GMP manufacturing and supports eventual IND, CTA, or commercial submissions.
OXB combines scientific expertise, platform technologies, and multi-vector experience to deliver reproducible, high-yield, and scalable viral vector processes. Our solutions minimize development risk, accelerate timelines, and ensure a smooth transition to GMP manufacturing, helping you bring therapies to clinic faster.


