Home – CDMO Solutions | Viral Vectors | Lentivirus
30 years of lentiviral vector manufacturing
As a pioneer in lentiviral vector technology, OXB brings three decades of proven experience to the development and manufacturing of lentiviral vectors for cell and gene therapies. Now a global CDMO, we support partners from early development through to commercialisation.
With over 340 GMP batches successfully manufactured, and 25 regulatory submissions supported, OXB offers deep regulatory, process, and analytical expertise to accelerate advanced therapy programmes from discovery to market.
OXB’s lentiviral vector track record
0+
GMP batches successfully released
0
Successful regulatory submissions
0
monthsTo GMP release
0E+
12 TUAt 200L scale
0%
Analytical assays performed in-house
0+
Patients’ doses
LentiVector™ platform
Taking novel therapies from discovery to commercialisation
OXB’s LentiVector™ platform offers a fully integrated and optimised suite of technologies and processes for the scalable production of high-quality lentiviral vectors. Designed to meet the complex demands of the cell and gene therapy market, the platform encompasses vector design, upstream and downstream processing, analytics, and fill/finish services, all aligned to deliver robust, reproducible GMP manufacturing.
The platform offers 3rd generation and 4th generation lentiviral vector systems, including proprietary innovations; 2KO genome™, SupA-LTR™, MaxPax™, and the TRiP System™, that together can enhance vector potency, productivity, quality, and safety.
With the LentiVector™ platform, clients benefit from high-yield, high-volume GMP manufacturing, scalable from early clinical to commercial supply, supported by deep regulatory expertise across global markets.
Explore the LentiVector™ platform
Our platform technologies:
Related resources
The TetraVecta™ System: Optimisation & Exemplification of OXB’s Next Generation Lentiviral Vector
View PDF >Development and Scale Up of Next Generation Lentiviral Vector Batch Process Demonstrating Increased Productivity and Enhanced Purity
View PDF >Increasing AAV Productivity Upstream Process Optimization for a Robust, Plug-and-Play Platform
View PDF >Why choose OXB as your LV CDMO?
Taking novel therapies from discovery to commercialisation
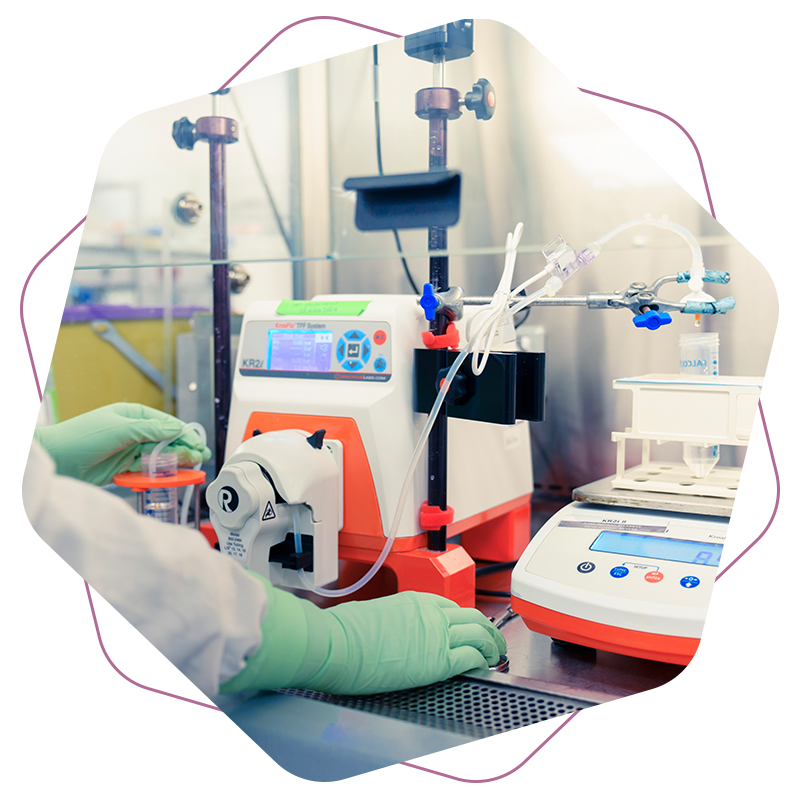
Access innovative technology and unmatched expertise
With decades of leadership in lentiviral vector production, OXB gives clients access to some of the industry’s most experienced scientific minds. Our commitment to continuous innovation ensures clients benefit from the latest advancements in viral vector manufacturing to deliver best in class cell and gene therapies.

Receive comprehensive regulatory support through to commercialisation
OXB provides end-to-end regulatory support, guiding clients from preclinical development through to commercial launch. We have successfully supported multiple IND and MAA submissions, and maintain a Drug Master File (DMF) with the FDA, reinforcing our credibility as a trusted global CDMO partner.

Leverage local manufacturing with a global footprint
With harmonised lentiviral vector manufacturing processes across our UK, US, and European facilities, OXB enables efficient, scalable production from 50L to 1000L, close to your clinical or commercial markets. Our global footprint ensures flexibility, consistency, and speed—wherever you are.
Ways to work with us
There are multiple ways in which pharma, biotechs and start-ups work with us, depending on development phase, scale, and individual IP requirements.
Platform project
For clients that are pre-clinical or early-clinical that wish to reach GMP at an accelerated rate with a proven process.
Custom solution
For clients that are pre-clinical or early-clinical that require flexibility in the manufacture of their drug candidate.
Tech-transfer
For clients in phase I, II, or III that need to transfer their manufacture to a CDMO for scale and/or long-term manufacture.



Frequently asked questions
Looking for something specific? – let's discuss a custom project
For pre-clinical clients that would like to co-develop a highly specific process, our custom project offering allows you to access OXB’s technology in combination with others to create a truly unique solution to meet your individual requirements.
